Tripledemic is the term that has been coined to describe a surge in cases of three viruses this fall, Covid 19, Respiratory Syncytial Virus and Influenza.
My phone has been ringing again. Not a good sign. There are new flu and Covid cases daily. Some people think that RSV just affects children. Not true. RSV increases the risk of hospitalization in older adults as well. I read the number of Covid 19 cases reported in the paper, but since most cases are determined with home testing, the official numbers are much lower. What really matters are the hospital cases and they are rising. This is an excerpt from an email sent to the staff of Methodist hospitals on Friday.
Date: December 2, 2022
Subject: MHS and COVID Updates
To: Methodist Healthcare Medical Staff
From: Paul Hancock, MD, Chief Medical Officer
Please note the following important MHS updates:
COVID Update
We are experiencing an increase in the number of COVID patients. MHS has 51 COVID positive patients under our care including 4 COVID patients in the ICU and 3 on ventilators.
MHS COVID Personal Protective Equipment (PPE) Guidance
Bexar, Atascosa, and Comal Counties – Transmission has increased to the HIGH level. While there are no changes in PPE guidance this week, if high transmission level is sustained for another week, we will move to universal masking in areas patients are encountered beginning December 12th.
Kendall County – Maintained SUBSTANTIAL or lower transmission. There are no changes to PPE guidance this week. More info
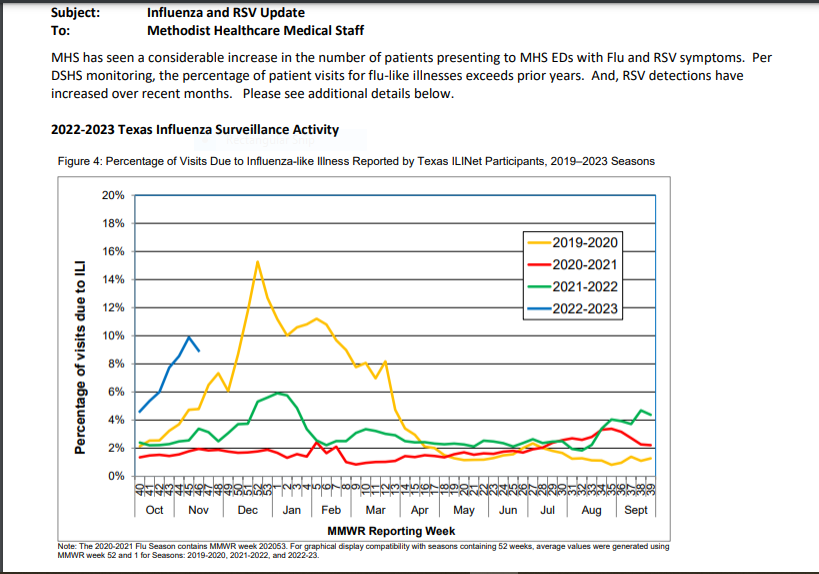
Influenza and RSV Update
MHS has seen a considerable increase in the number of patients presenting to MHS EDs with Flu and RSV symptoms. Per DSHS monitoring, the percentage of patient visits for flu-like illnesses exceeds prior years. And, RSV detections have increased over recent months. More info
One other bit of bad news. Monoclonal antibodies which are used to treat Covid 19 are no longer effective against newer variants. I suspected this given a recent experience with a patient who couldn’t take Paxlovid because of some of the medications that he was taking and a concern about drug interactions. I arranged for a home infusion of bebtelovimab and it didn’t seem to expedite his recovery.
FDA Pulls Authorization for Bebtelovimab Due to Nonsusceptible SARS-CoV-2 Variants

Bebtelovimab is currently not authorized for emergency use to treat COVID-19 in any US state or territory at this time, according to an updated alert from the Food and Drug Administration (FDA).
The latest data from the Centers for Disease Control and Prevention show that the SARS-CoV-2 variants BQ.1 and BQ.1.1 account for more than 57% of COVID-19 cases nationally, and continue to be increasingly prevalent across all US regions. Neutralization data indicate that bebtelovimab is not expected to neutralize these variants and as such is no longer authorized for use at this time. The updated information can be found in the Fact Sheet for Health Care Providers.
Health care providers are urged to consider alternative therapies such as Paxlovid, Veklury (remdesivir), and Lagevrio (molnupiravir), which are expected to work against the BQ.1 and BQ.1.1 subvariants. These treatments are authorized or approved to treat patients with mild to moderate COVID-19 who are at high risk for progression to severe disease, including hospitalization or death.
The commercial distribution of bebtelovimab has been paused by Eli Lilly and its authorized distributors until further notice. Product that has already been distributed should be retained in the event that SARS-CoV-2 variants susceptible to bebtelovimab become more prevalent in the future in the US.
Reference
FDA announces bebtelovimab is not currently authorized in any US region. News release. US Food and Drug Administration. Accessed November 30, 2022. https://www.fda.gov/drugs/drug-safety-and-availability/fda-announces-bebtelovimab-not-currently-authorized-any-us-region
BOTTOM LINE: GET A FLU SHOT. GET A BIVALENT BOOSTER. WEAR A GOOD MASK WHEN YOU ARE IN CLOSE QUARTERS IN CROWDS. WASH YOUR HANDS AND, MOST IMPORTANTLY, STAY HOME IF YOU ARE SICK.