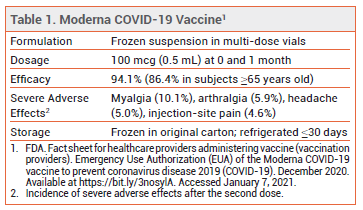
I don’t have any new tips on getting the vaccine, but one positive development is that those patients who received their vaccinations today at the Alamodome received follow up appointments for their second dose. Patients who did receive them Monday and Tuesday will be contacted.
Johnson and Johnson’s vaccine showed promising results in a study published today in the New England Journal of Medicine. This is a summary from Journal Watch followed by the abstract from today’s Journal. The results of a clinical study to show it’s efficacy will be released at the end of the month. If those results are positive, it will be considered by the FDA for Emergency Use Authorization.
By Kelly Young
Edited by Susan Sadoughi, MD, and Richard Saitz, MD, MPH, FACP, DFASAM
A single dose of Johnson & Johnson’s Ad26.COV2.S vaccine appears to be safe and induce an immune response in two age groups, according to interim results of a phase 1-2a trial published in the New England Journal of Medicine.
A total of 800 adults aged 18 to 55 or aged 65 and up were randomized to various combinations of low-dose or high-dose vaccines or placebo, given 56 days apart.
Adverse events were common, with fatigue, headache, myalgia, and injection-site pain reported most often. At day 29 after the first dose, the seroconversion rate was 99% or more in the younger cohort across dosing groups. Older vaccine recipients had a 96% seroconversion rate. At 57 days after the first dose, antibody titers had increased further.
Interim Results of a Phase 1–2a Trial of Ad26.COV2.S Covid-19 Vaccine